10 min read AlphaFold Protein structures are defined, experimentally, by X-ray crystallography, nuclear magnetic resonance and electron cryomicroscopy (cryo-EM). However, these techniques are complex, time consuming, expensive and the protein is often not in its native form (Bertoline et al., 2023). Therefore, from its introduction in 2018, DeepMind’s AlphaFold program has […]
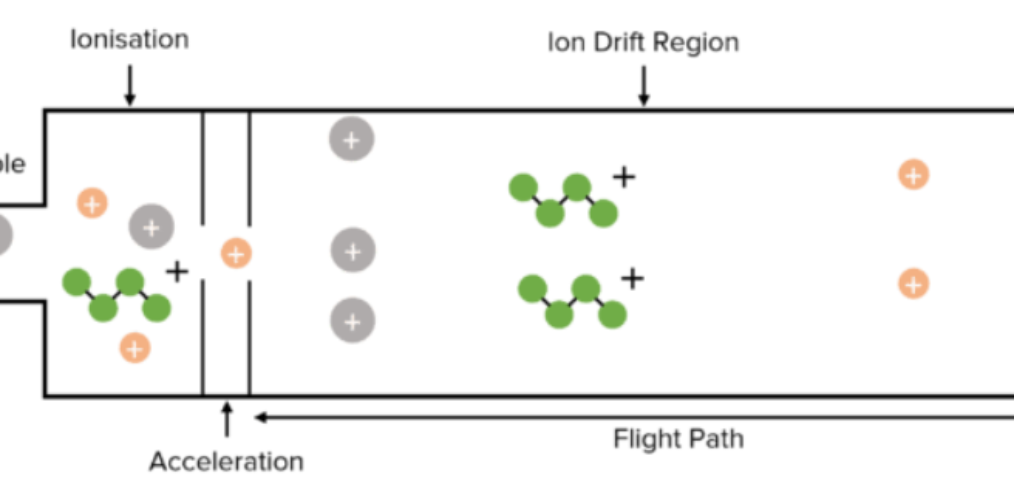
15 min read. Basic principle: Time of flight mass analysers use an electric field to accelerate ions along a field-free drift path of known length, and then measures the time each ion takes to reach the detector. This is shown in the diagram below. Time of flight (ToF) is dependent on the mass to charge […]
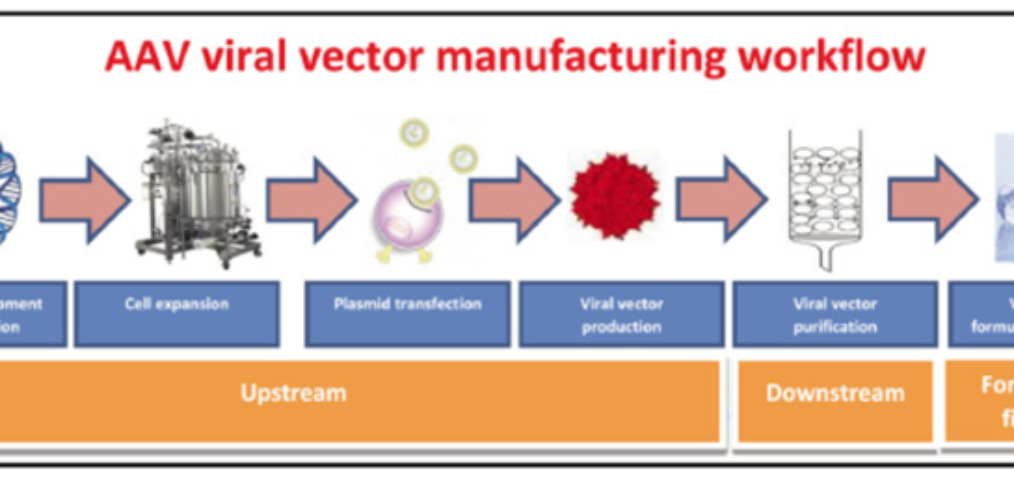
20 min read. The adeno-associated virus (AAV) was first discovered in 1965 as a contaminant of adenovirus preparations, subsequently AAV was found in human tissues. Pure scientific curiosity in understanding basic AAV biology is what led to realising the tremendous potential of AAV as a vector in human gene therapies. AAV vectors are now the […]
15 min read. Chemotherapeutic agents used to treat cancer require accurate dosing to prevent serious adverse toxicities. For those drugs excreted through the kidney, a precise understanding of kidney function (as measured by glomerular filtration rate, GFR) is needed to ensure drug toxicity is avoided. Carboplatin is unique among chemotherapeutic agents in that its dosing […]
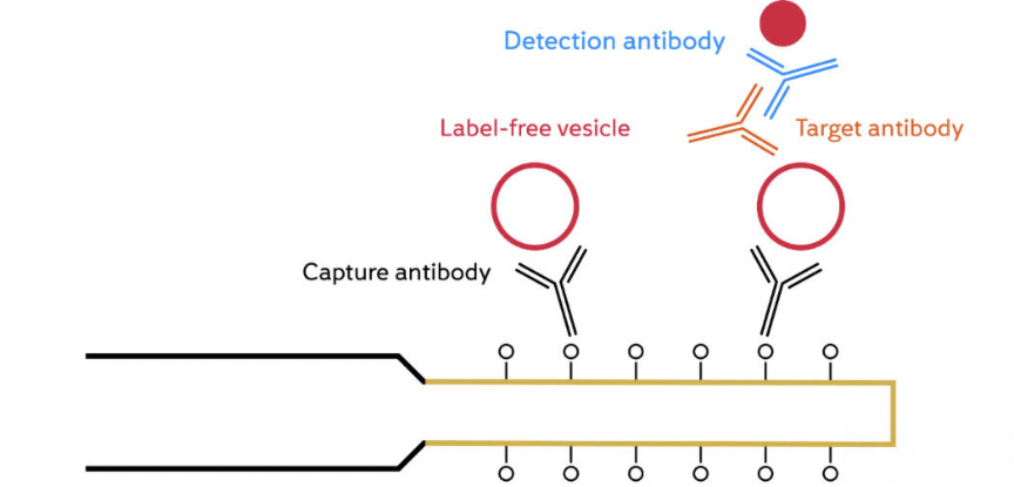
Extracellular vesicles (EVs) play a crucial role in mediating cell-to-cell communication. They are involved in several physiological and pathological processes within the body, including cancer development, and potential use as non-invasive cancer biomarkers. However, the extensive heterogeneity in size, composition, and origin of EVs makes their accurate and reliable characterization and detection crucial to meet […]
Publishing a research paper is a significant milestone in any academic or professional career. It’s an achievement that requires hard work and dedication. We wanted to take a moment to acknowledge our lab manager’s Dr. Carolyn Hyde, amazing work on this recent research paper, “Domestic Animal Endocrinology”. Let’s take a summarised dive into this blog […]
Atypical myopathy in horses is a severe and frequently lethal equine disease. Its cause is suspected to be the ingestion of a toxin and it develops sporadically. This illness causes a muscle disorder that progresses rapidly, with the characteristic of degeneration and necrosis of postural, respiratory and lingual muscles. The word “atypical” represents the fact that it […]
What is it? By filtering molecules over a gel, size exclusion chromatography (SEC) separates molecules according to their size (1). The gel is made up of sphere-shaped beads with pores that vary in size (1). When molecules of various sizes are passed through the gel, separation occurs because the smaller molecules can enter the pores […]
Incorrect Labelling If the labelling is done incorrectly and samples are mixed up as a result, the only thing we can do is repeat the whole experiment, wasting time and products. When using a handwritten label, make sure it is authentic; always add the date of labelling, the expiry date and your initials. […]
The identification, measurement, and purification of a specific analyte or chemical are the primary goals of the Chromatographic process. Examinations can be done both quantitatively and qualitatively. HPLC is just one aspect and can be used in the following applications (1): Purifying water Impurity detection in the pharmaceutical sectors Trace component pre-concentration Ligand-exchange Ion-exchange of […]