Size Exclusion Chromatography
What is it?
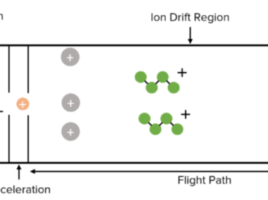
By filtering molecules over a gel, size exclusion chromatography (SEC) separates molecules according to their size (1). The gel is made up of sphere-shaped beads with pores that vary in size (1). When molecules of various sizes are passed through the gel, separation occurs because the smaller molecules can enter the pores and the larger ones cannot (1). Large molecules are eluted in the column’s void volume, whereas small molecules diffuse into the pores and flow through more of the column in proportion to their size (1). As a result, as molecules go through the column, they segregate based on their sizes and are eluted in decreasing size or molecular weight order (1).
Separation
Desalting – Desalting samples of proteins or nucleic acids is a typical application for SEC. Smaller molecules are held in the gel pores while the important molecule is eluted in the void volume (1). The gel should have an exclusion limit that is much smaller than the target molecule in order to achieve the desired separation (1).
Fractionation – Within the gel matrix, molecules with various molecular weights are separated (1). The compounds of interest should fit inside the gel’s fractionation range when using this separation technique (1).
How does SEC work?
SEC resins are made up of spherical particles (beads) that are neither reactive nor adsorptive and that contain pores (2). Molecules larger than the pores cannot diffuse into the beads when the sample enters the column, therefore they will elute first (2). Depending on their size, molecules of a size between the very huge and the very small can infiltrate the pores to varied degrees (2). A molecule will be able to penetrate the total pore volume if it is smaller than the resin’s smallest pore (2). The last molecules to elute are those that enter the entire pore volume. There is no need to utilise various buffers during the separation because samples are isocratically eluted (2). These resins are fragile in comparison to silica based packing materials, so the chromatography is usually done at low flow rates so as not to incur too much pressure.
The range of molecular weights (MW) that can be separated and have partial access to the pores of the particle is defined by the intended fractionation range of the SEC resins used for the purification of biomolecules (2). Peptides and other tiny biomolecules can be separated using fractionation ranges of 100 to 7000 MW, whereas big proteins like antibodies can be purified using resins with fractionation ranges of 100 000 to 300 000 MW (2). The size of the molecules that are excluded from the pores and subsequently elute in the void volume is indicated by the resin’s exclusion limit (2).
How to optimise separation
The main factor determining the performance of SEC is the selection of circumstances that provide adequate selectivity and mitigate peak broadening effects during the separation. In most cases, prepacked columns are given with suggested running settings that produce good results, but tuning may occasionally be required to achieve the desired resolution. Resolution is a result of the selectivity of the resin and the effectiveness of that resin to produce narrow peaks.
If optimization is required, follow these steps:
- Choose a resin with a fractionation range that will provide the best resolution.
- Choose a column whose bed height offers the necessary resolution. It is advised that beds be between 30 and 100 cm high for preparative separation.
- Depending on the amount of sample that has to be processed, choose a column size that is adequate.
- Choose the maximum flow rate that minimises separation time while maintaining resolution.
References
- BIO-RAD. Introduction to Size Exclusion Chromatography. (Internet) (Cited on 2022 September) Available from: https://www.bio-rad.com/en-uk/applications-technologies/introduction-size-exclusion-chromatography?ID=MWHAXJKG4
- Cytiva. Fundamentals of size exclusion chromatography. (Internet) (Cited on 2022 September) Available from: https://www.cytivalifesciences.com/en/us/solutions/protein-research/knowledge-center/protein-purification-methods/size-exclusion-chromatography