Polyaminopropyl Biguanide (PHMB) is a broad-spectrum antimicrobial compound found in contact lens cleaning solutions, skin disinfection treatments, and wound dressings, among other things (1). However, this substance has been banned in cosmetics in Europe since 2014. Products such as makeup, creams and body lotions used to contain PHMB as a preservative (2), but PHMB is […]
Lactulose Lactulose is a non-absorbable synthetic disaccharide made up of galactose and fructose that is also known as 1,4 beta galactoside-fructose (1). Because the human small intestinal mucosa lacks the enzymes needed to divide lactulose, it reaches the large intestine intact (1). Colonic bacteria convert lactulose to monosaccharides, which are then converted to volatile fatty […]
Symmetric dimethylarginine (SDMA) and its isomer, asymmetric dimethylarginine (ADMA) are amino acid derivatives which inhibit the body’s ability to produce nitric oxide, a molecule that aids in the maintenance of a healthy endothelium. Moreover, these compounds are formed by intranuclear methylation of L-arginine residues on various regulatory proteins and are released into the cytoplasm following […]
What are bile salts? Bile salts are a primary component of bile, which is a yellowish-green fluid produced by the liver and stored in the gallbladder. Function of bile salts Bile salts aid in fat digestion, elimination of toxins and absorption of fat-soluble vitamins such as A, D, E, and K (1). How are they […]
What is Nicotinamide? Nicotinamide (also known as Niacinamide) is a form of vitamin B3, which is necessary for the body’s processing of lipids, amino acids, and glucose. A deficiency can have an impact on appetite as well as red blood cell production. Vitamin B3 is an essential vitamin for cats, because they cannot produce it […]
The use of pesticides The agriculture industry faces pressure to protect yields and crops to ensure they can keep up with the high demand and produce large amounts of food. To do this they need to use pesticides (fig.1). Pesticides are used to protect agricultural production and planting from insects, weeds, fungi and other pests. […]
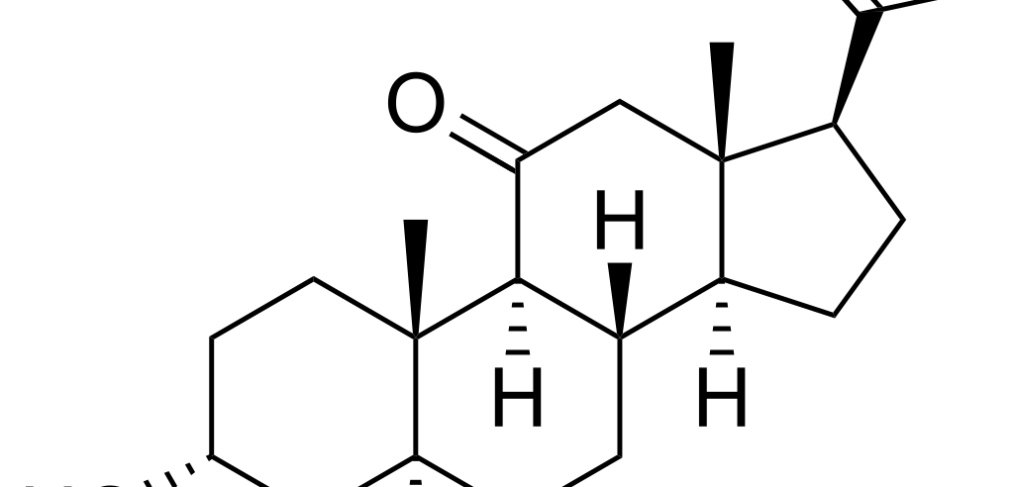
What’s Alfaxalone and its function Alfaxalone (Fig.1) is a neuroactive steroid that has anesthetic properties by acting on the gamma-aminobutyric acid subtype A (GABAA) receptors. It can act as a GABA agonist, when administered in high concentrations or modulate the chloride ion transport across the membrane of neuronal cells when given at low concentrations. Alfaxalone […]
Mannitol is an osmotic diuretic that is physiologically inactive in humans and is found naturally in fruits and vegetables as a sugar or sugar alcohol. Mannitol raises the osmolality of blood plasma, allowing more water to move from tissues including the brain and cerebrospinal fluid into interstitial fluid and plasma (1). Mannitol can be used […]
Amlodipine has been used to treat high blood pressure in humans, but it has now been approved for veterinary practice. It is given to cats and dogs to treat hypertension and other associated diseases. About Amlodipine This drug is classified as a calcium channel blocker. The heart and arteries contract more forcefully when calcium is present, thus […]
What is Pregabalin? “Pregabalin is used to treat epilepsy and anxiety, it is also taken to treat nerve pain that can be caused by different illnesses including diabetes, shingles, or injury. Pregabalin works in different ways: 1) in epilepsy it stops seizures by reducing the abnormal electrical activity in the brain 2) with nerve pain […]